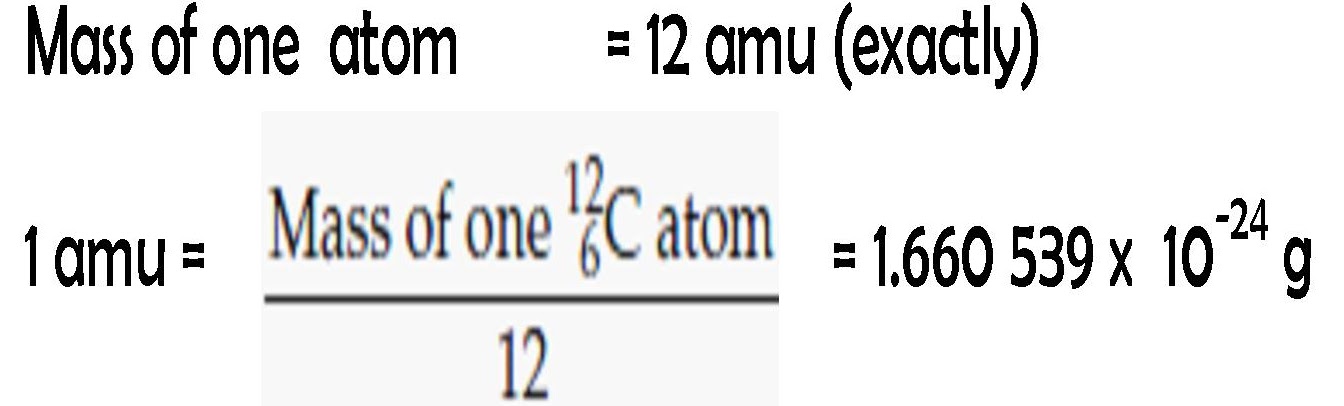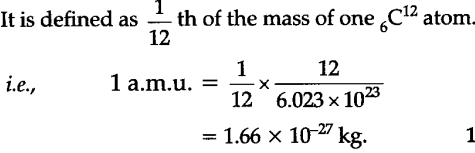16. If the atomic mass unit 'u' were defined to be of the mass of an atom of C-12. what would be the atomic weight of nitrogen in amu or 'u' in

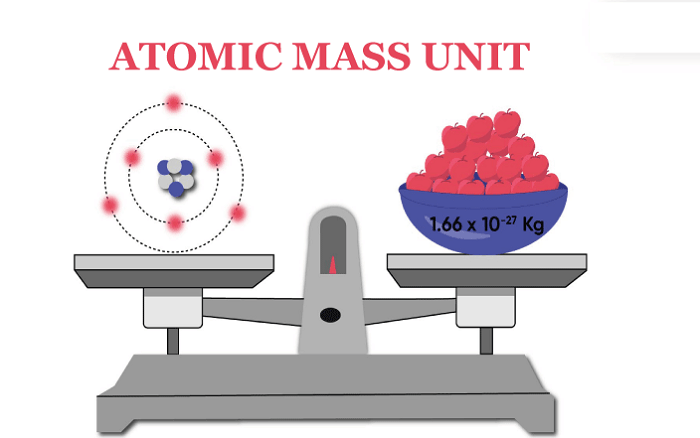
STRUCTUREThe mass of an atom or molecule is measured in atomic mass units. Which is greater, a carat or a milligram? Explain.

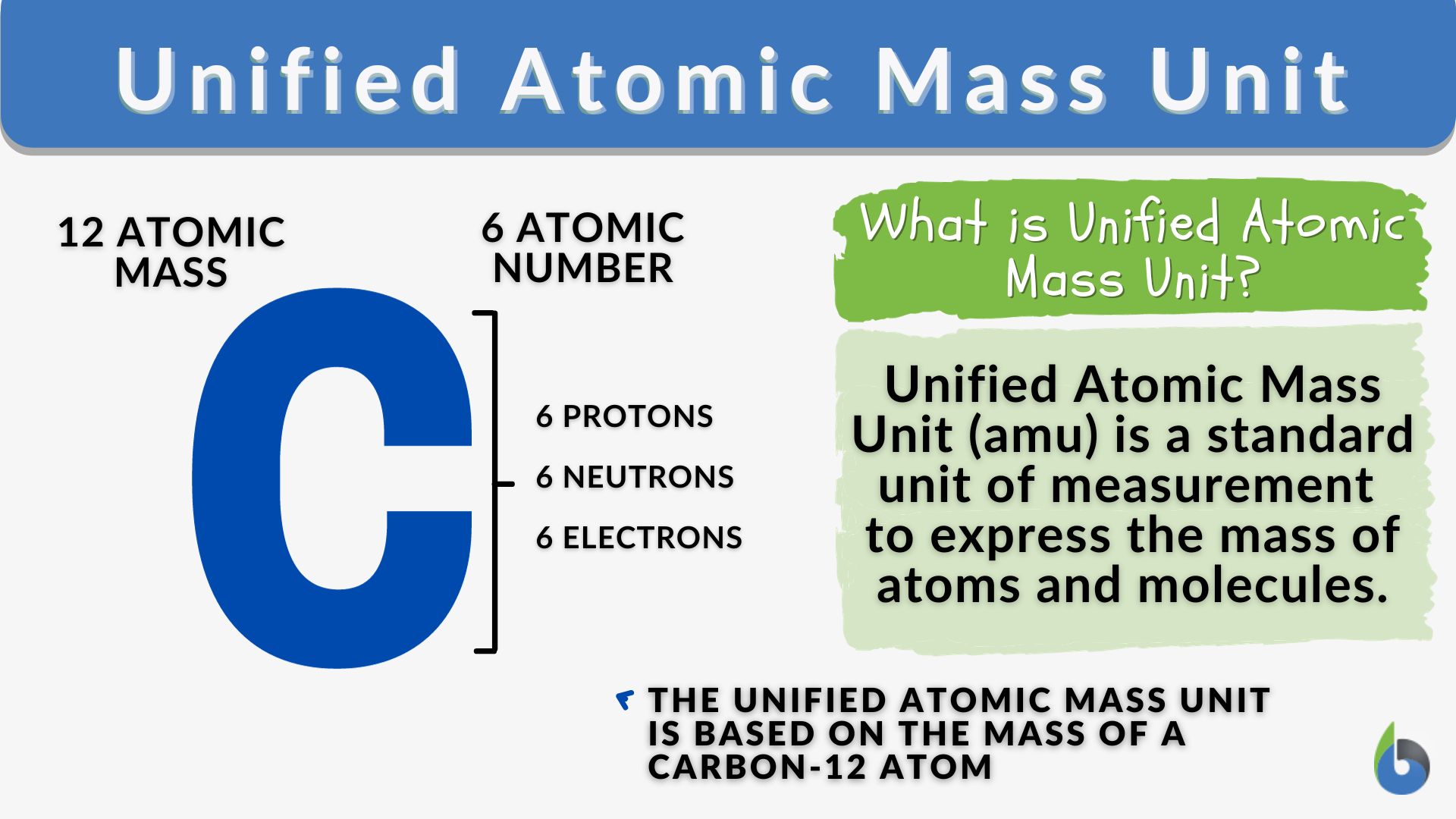
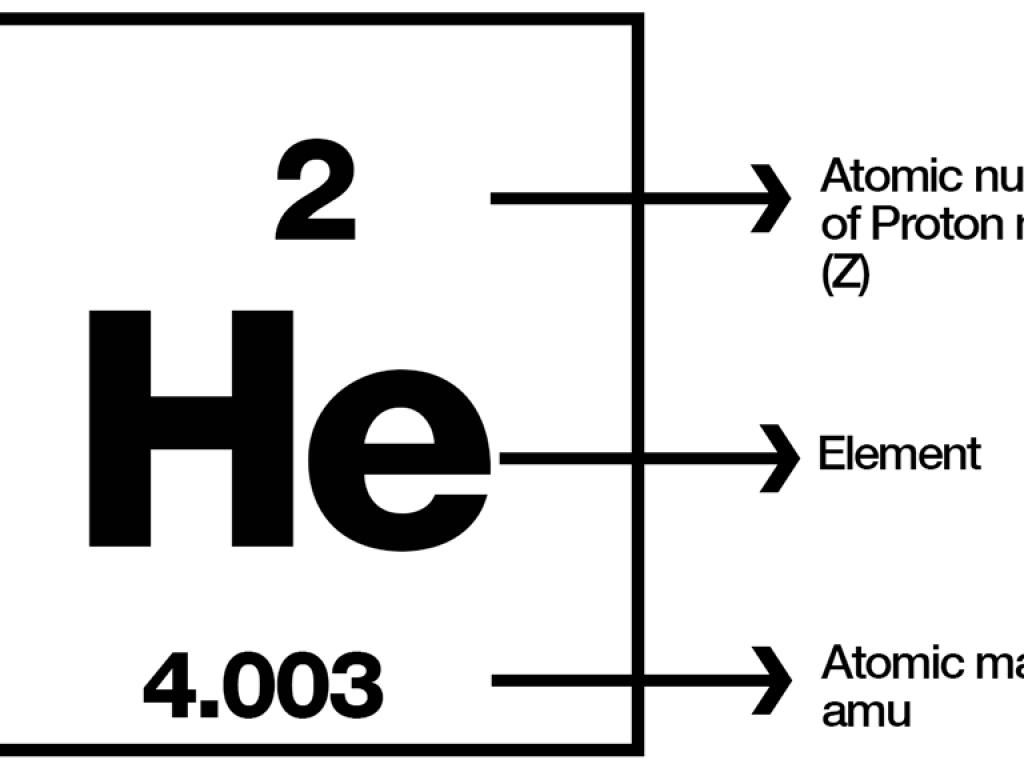
Moles Notes. 1. Atomic Mass Unit amu – atomic mass unit, used to describe the mass of an atom Conversion factor: 1 amu = 1.66 x 10 -24 g Equivalence statement: - ppt download

on IU - 1.0005 X 10 kg. 9. What is the meaning of 'atomic mass unit? Calculate the equivalent energy in MeV of a unified atomic mass unit. (ISC 2007) 14. Th no n
What are the units of unified atomic mass units (nuclear physics, atomic physics, units, physics)? - Quora