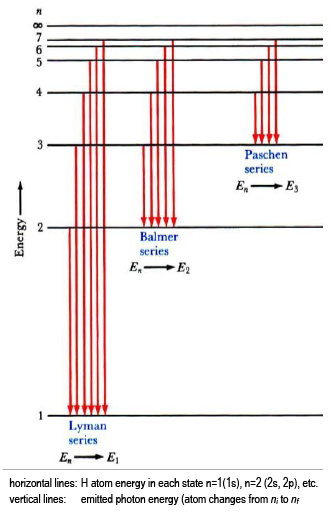
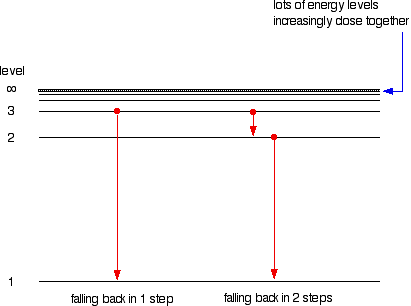
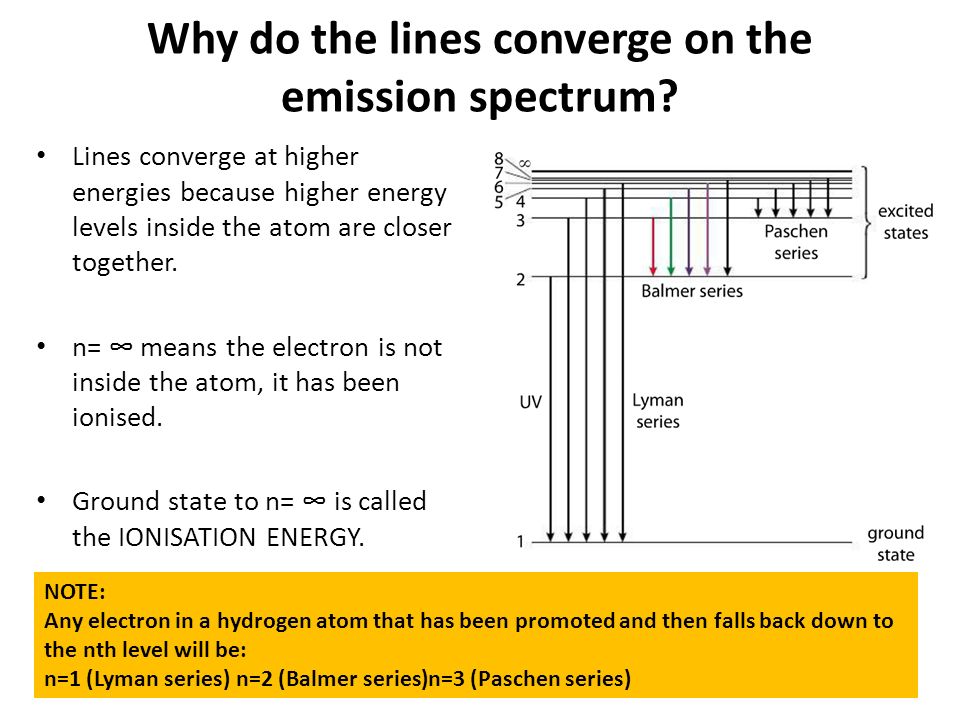
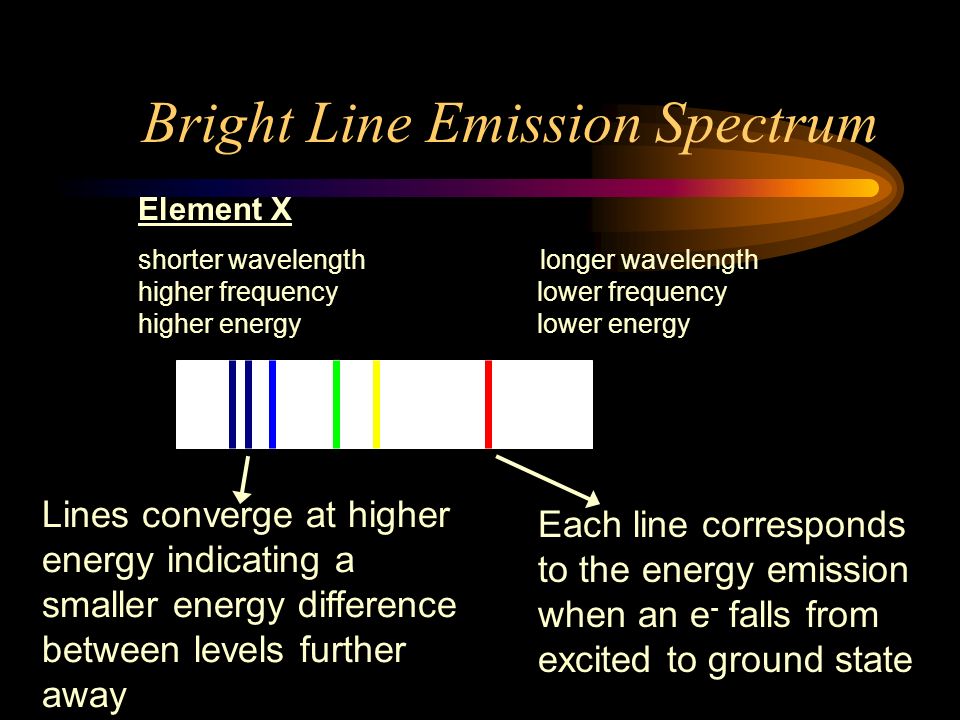
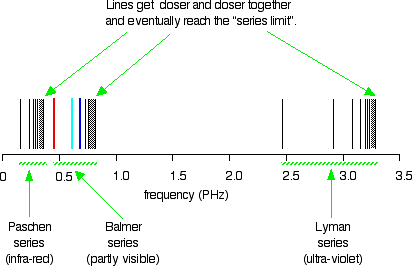
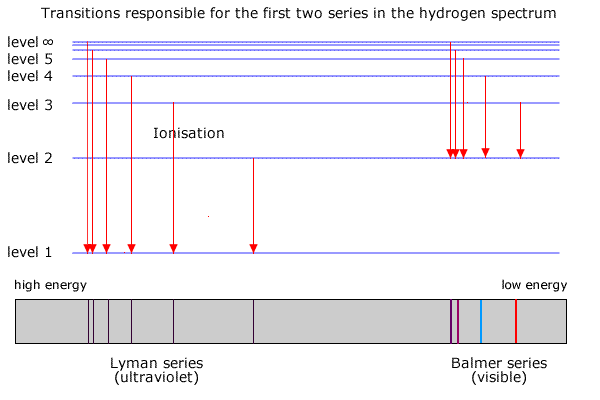
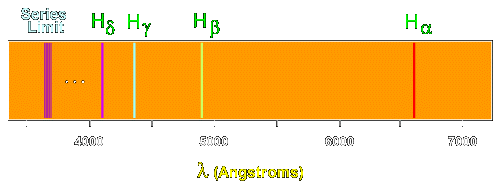
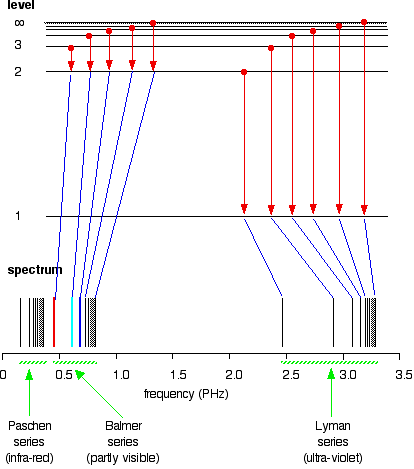
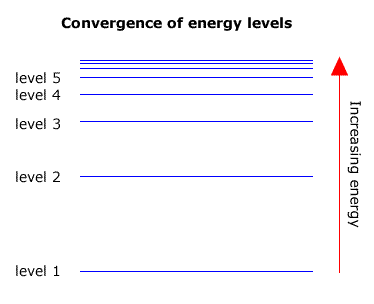
6. The line emission spectrum of hydrogen provides evidence for the existence of electrons in discrete energy levels, which converge at higher energies. – The Atomic Project (SL Chemistry)

Hydrogen emission spectrum spectroscopy successive ionisation energy patterns related to sub-shells and group of periodic table GCE A Level revision notes

Emission Spectra & First Ionisation Energy (12.1.1) | DP IB Chemistry: HL Revision Notes 2016 | Save My Exams

Me Structure 106. Which statement relating to the spectrum of H atom is false : (A) The lines can be defined by quantum number (B) The lines of longest wavelength in the
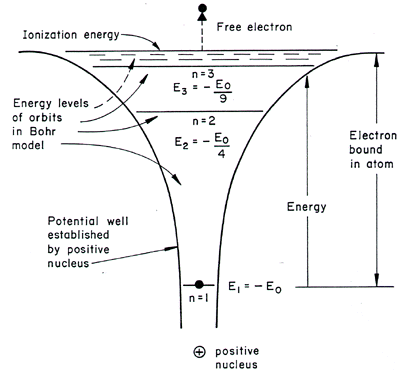
Why are the convergence lines in the UV spectrum used to calculate the ionization energy of hydrogen rather than the convergence line in the visible spectrum? | Socratic

Since an atom has infinity energy levels, why does an emission spectrum for a hydrogen atom for example, have very few emission lines? - Quora
HOW does the line emission spectrum of hydrogen provide evidence for the existence of electrons in discrete energy levels, which converge at higher energies? - Quora

6. The line emission spectrum of hydrogen provides evidence for the existence of electrons in discrete energy levels, which converge at higher energies. – The Atomic Project (SL Chemistry)