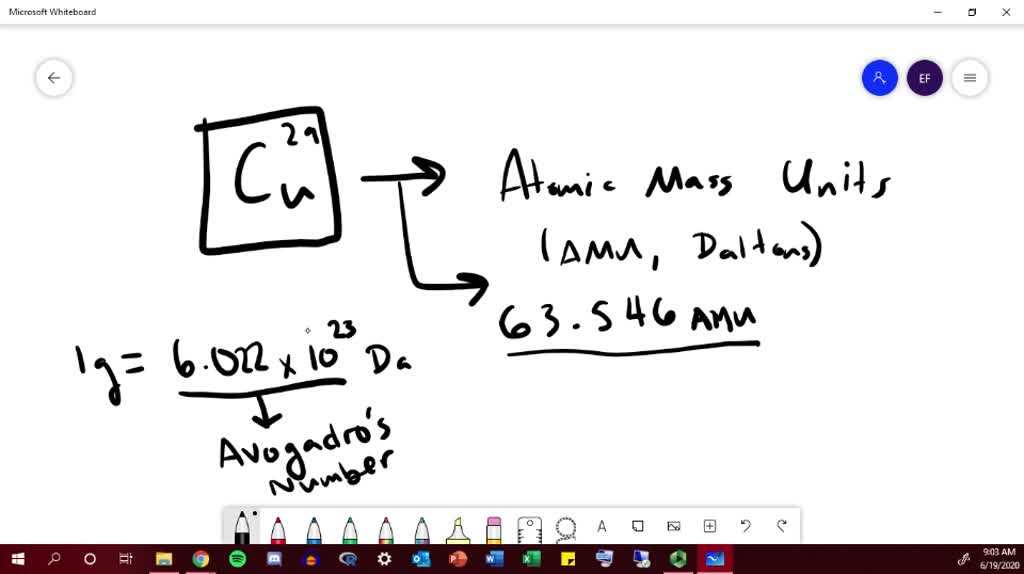
Average Atomic Mass. An element can exist in a number of forms, called isotopes. Isotopes are forms of the same atom that vary in mass as a result of. - ppt download

Calculate mass of `Cu` in `3.67 xx 10^(3)g CuFeS_(2)` ? (Atomic mass `Cu = 63.5 Fe = 56,S = 32)` . - YouTube

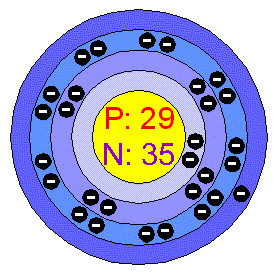
What is the mass of one atom of copper? How many copper atoms are in 3 cm^3 of copper? How many copper atoms are in 5 g of copper? Niobium at 20

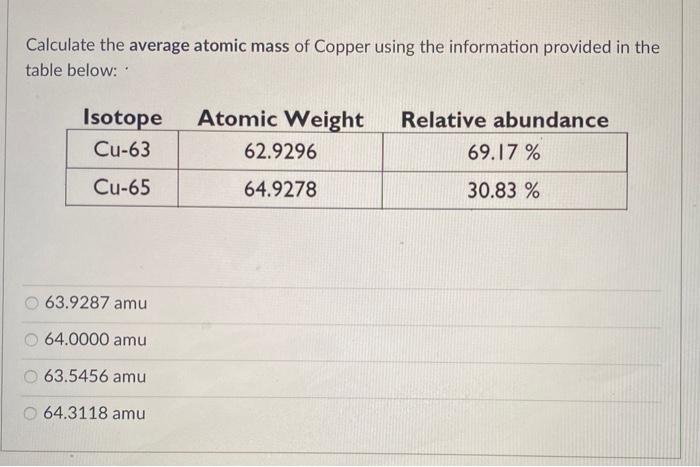
the of a diagram. Naturally occuring copper consists of isotopes of , and Cu,, in the ratio of 8:3. Calculate the average atomic mass of copper.

that the result is in agreement with law of multiple proportions. Q11. The average atomic mass of copper is 63.5. It exists as two isotopes 29 Cu and 29Cu. Calculate the percentage