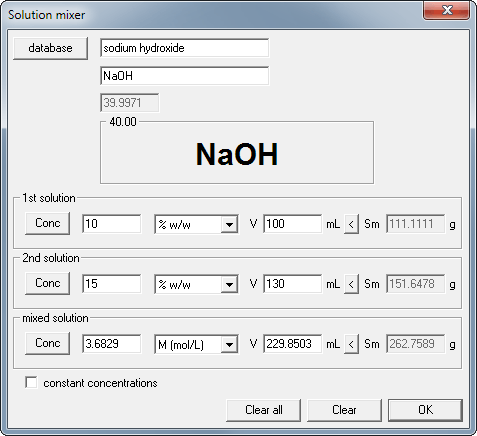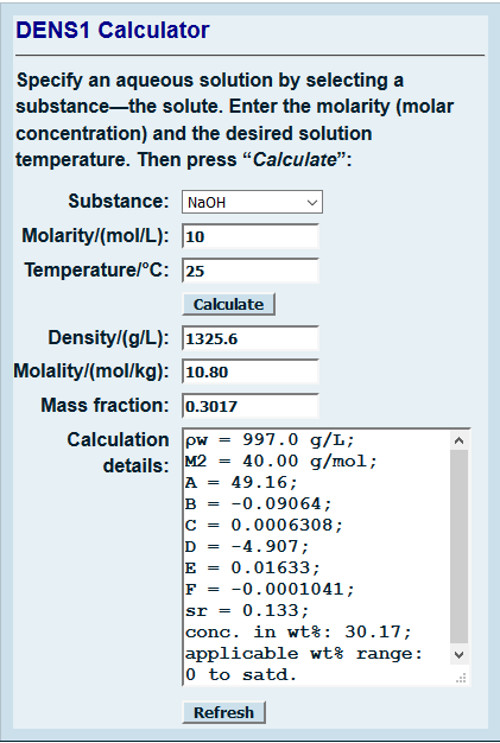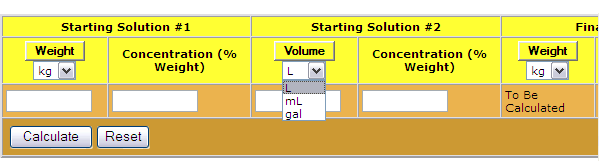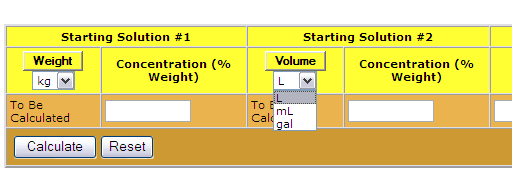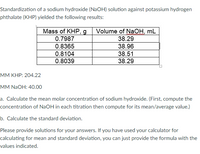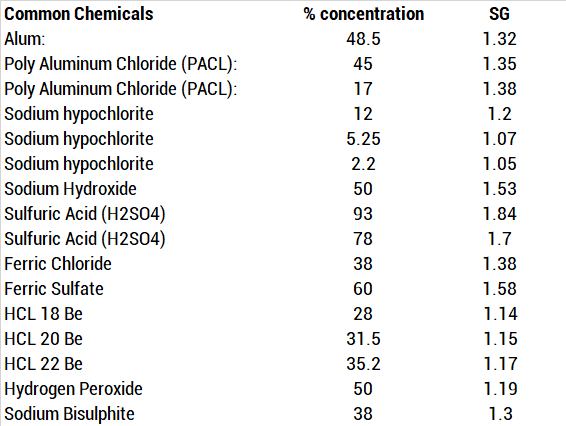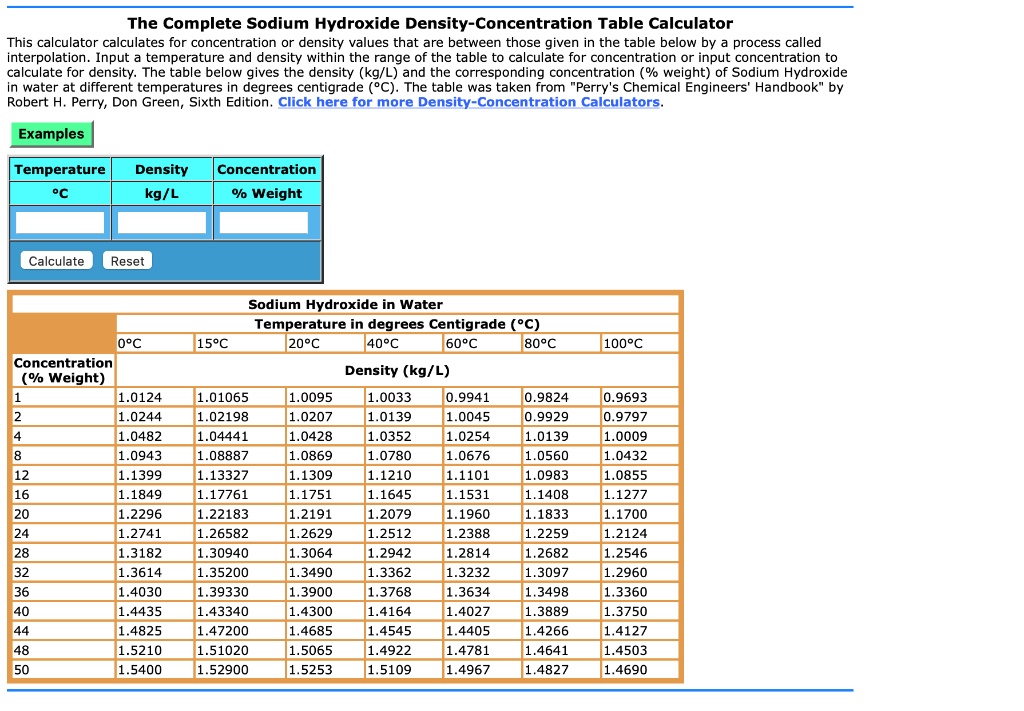
SOLVED: The Complete Sodium Hydroxide Density-Concentration Table Calculator This calculator calculates concentration or density values that are between those given in the table below by a process called interpolation. Input temperature and

Premium Photo | Double exposure of abstract virtual chemistry hologram on blurry calculator with laptop background research and development concept

SPEX CertiPrep - SPEX CertiPrep IS-BUF2-500 10M Sodium Hydroxide (NaOH) Ionic Strength Adjustment Buffer, 10 M, 500 mL